Clinical Trials & Pipeline
Clinical Trials & Pipeline
At Bayer, our focus is to discover and develop medicines that provide the opportunity to live a healthier life. We are actively pursuing our vision to deliver best-in-class patient care, outcomes, and experience by advancing science.
Information on investigational compounds and clinical trials sponsored by Bayer is made available here to increase the transparency of Bayer's clinical research. Below you can learn more about trials sponsored by Bayer. Research sponsored by independent investigators is not included.
The compounds presented are investigational or are being investigated for uses that have not been approved by the US Food and Drug Administration (FDA),
European Medicines Agency (EMA), or other health authorities. This information is presented only for purposes of providing a general overview of clinical trials and should not be construed as a recommendation for use of any product for unapproved uses.

OCEANIC-STROKE
A Study to Test Asundexian for Preventing a Stroke Caused by a Clot in Participants After an Acute Ischemic Stroke or After a High-risk Transient Ischemic Attack, a So-called Mini Stroke
STUDY DESIGN
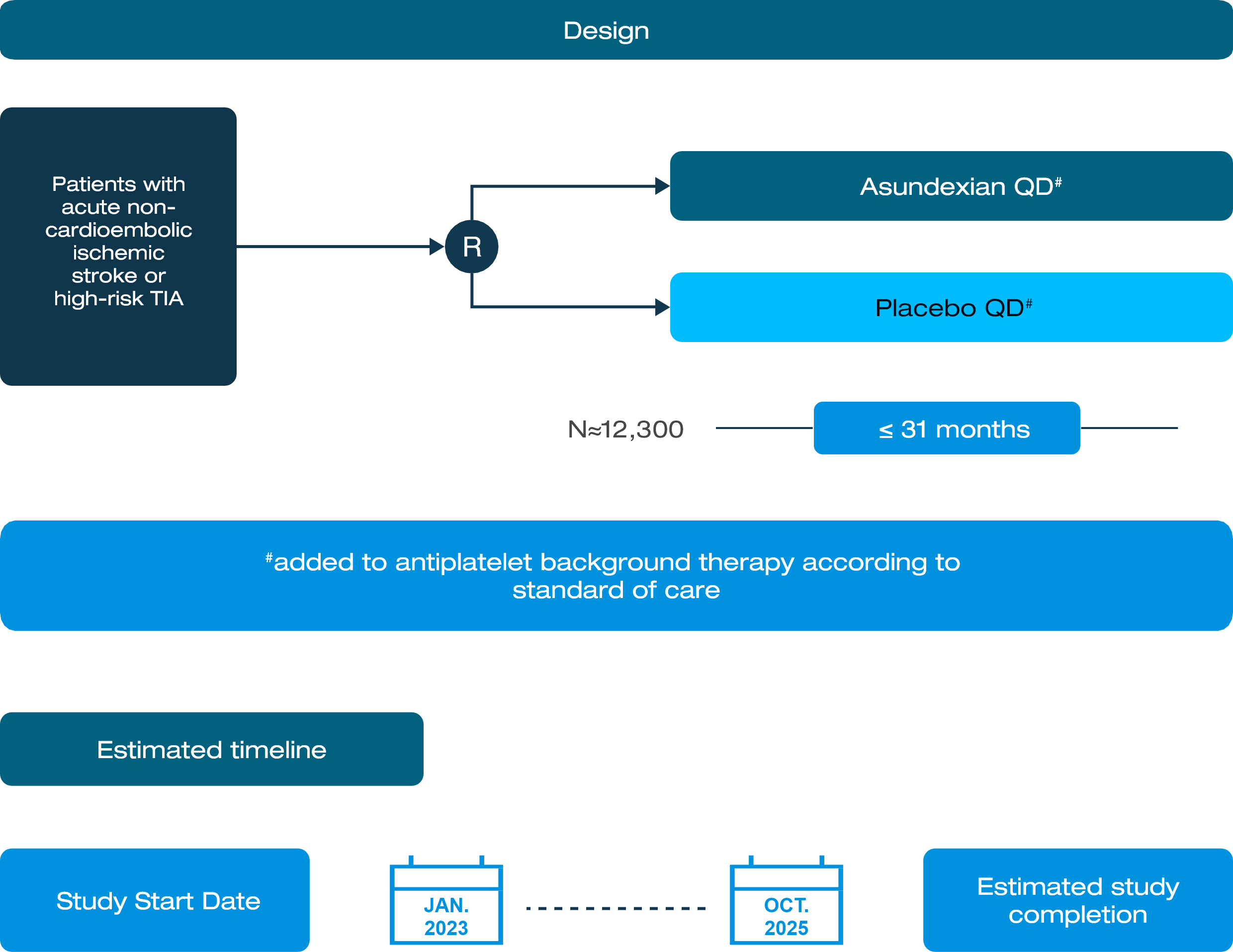
Inclusion Criteria:
- Participants must be ≥18 years of age
- Acute non-cardioembolic stroke or high-risk TIA
- Systemic or cerebrovascular atherosclerosis or acute non-lacunar infarct
Exclusion Criteria:
- Ischemic stroke ≤7 days before the index event
- Index stroke following procedures or strokes due to other rare causes
- History of atrial fibrillation/flutter, left ventricular thrombus, mechanic valve or other cardioembolic source of stroke requiring anticoagulation
Primary Efficacy Endpoint
Ischemic stroke
(Time to first occurrence)
Primary Safety Endpoint
ISTH major bleeding
(Time to first occurrence)
Secondary Endpoints
All strokes (ischemic and hemorrhagic)
Composite of CV death, MI, or stroke
Composite of all-cause mortality, MI, or stroke
Disabling stroke (modified Rankin Scale ≥ 3 at 90 days)
All-cause mortality
TIA
Composite of ISTH major or clinically relevant non-major bleeding
ISTH clinically relevant non-major bleeding
Symptomatic intracranial hemorrhage, hemorrhagic stroke
Fatal bleeding
Minor bleeding
Ischemic stroke or ISTH major bleeding
Composite of CV death, all stroke, MI, or ISTH major bleeding
Composite of all-cause mortality, disabling stroke, fatal bleeding, or symptomatic intracranial hemorrhage (Time to first occurrence)
NCT06728332
Acronyms:
CV: Cardiovascular, ISTH: International Society on Thrombosis and Hemostasis, MI: Myocardial infarction, mRS: Modified Rankin Scale, TIA: Transient Ischemic Attack


